Sometimes, hydrogen is also produced during cracking. 
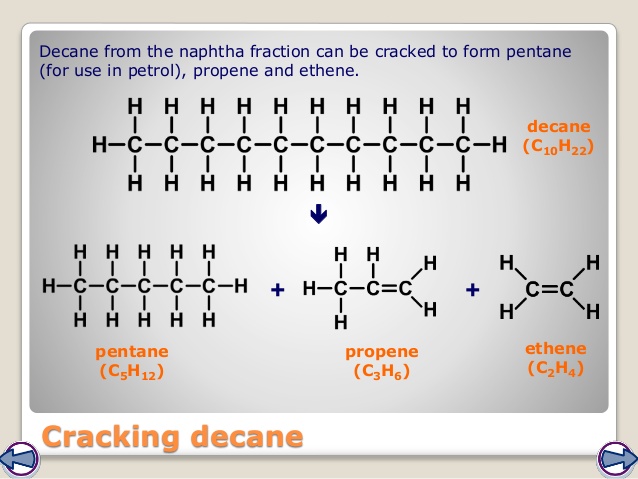
Some of the smaller hydrocarbons formed by cracking are used as fuels (eg large chains are often cracked to form octane for petrol, which is in high demand), and the alkenes are used to make polymers in the manufacturing of plastics. There is a greater demand for smaller hydrocarbons than larger ones. Cracking produces smaller alkanes and alkenes. These processes break covalent bonds in the molecules, causing thermal decomposition reactions. 2 C10H22 + 21 O2 > 20CO + 22H2O What is the balanced chemical equation of the combustion of decane 2C10H22 + 31O2 20CO2 + 22H2O What is the chemical. passed over a catalyst of silica or alumina.Fractions containing large hydrocarbon molecules are heated to vaporise them. This is where cracking comes in.Ĭracking allows large hydrocarbon molecules to be broken down into smaller, more useful hydrocarbon molecules. (ii) Write an equation for the cracking of one molecule of dodecane into. Crude oil often contains too many large hydrocarbon molecules and not enough small hydrocarbon molecules to meet demand. There is a greater demand for smaller hydrocarbons than larger ones. (i) Give the general formula for the homologous series that contains dodecane. Fuels made from oil mixtures containing large hydrocarbon molecules are not efficient as they do not flow easily and are difficult to ignite.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
